- Promo !
- -130,00 €







411,67 €
Pack Economique
Across Revolax 10 BOITES
Acide hyaluronique injectable
10 seringues de 1,1mL
 Vous nous avez noté !
Vous nous avez noté !

 Paiement 100% sécurisé
Paiement 100% sécurisé
Tous vos paiements sont sécurisés. Un doute ? Contactez nous !
 Livraison Express
Livraison Express
Nos solutions de livraisons sont très rapides, Expédition le jour de votre commande. DHL 24H
 Service client réactif
Service client réactif
Via téléphone, Chat, SMS, e-mail N'hésitez pas à poser vos questions à notre équipe de pharmaciens.
Grâce à sa capacité avancée à modeler, à maintenir sa structure et à sa longévité, il est recommandé pour le traitement des rides profondes à graves, y compris les nasolabiales et les contours du visage (joue, menton ou nez).
Composition
Acide Hyaluronique : 24 mg / ml
Lidocaïne HCl 0,3%
Volume 1.1ml
Durée 12 à 18 mois
Across est un laboratoire Coréen.
Il bénéficie du marquage CE.
REVOLAX :
- Acide hyaluronique de haute pureté Non-animal
- Substances naturelles extraites de bactéries
- Haute pureté (endotoxine <0,0015 UI / mg)
- Biodégradable (absorption naturelle)
- Forte réticulation : Technologie de réticulation fine d'Across
- Amélioration de la durabilité
- Monophasique hautement cohérent
- Motif régulier et dense
- Structure de gel stable et constante
- Effet naturel/ Injection douce
- Haute viscoélasticité
- Intensifier la persistance de durabilité plus longue
- Maintient l'élasticité en soutenant fermement le tissu cutané
Pourquoi utilisez-vous Revolax ?
FICHE TECHNIQUE DE REVOLAX SUBQ
DESCRIPTION
REVOLAX SUB-Q lidocaïne est un gel physiologique stérile sans pyrogène d'acide hyaluronique réticulé d'origine non animale avec
l'ajout de 0,3% de chlorhydrate de lidocaïne. Il s'agit d'un gel aqueux incolore, inodore et très
visqueux.
REVOLAX SUB-Q lidocaïne est présenté sous la forme d'un volume stérile de 1,1 ml dans une seringue en verre à usage unique, accompagné
de deux aiguilles stérilisées.
INDICATIONS
REVOLAX SUB-Q Lidocaïne est indiqué pour l'implantation intradermique à la perte de volume ainsi que pour l'asymétrie morphologique
du visage. L'ajout de la lidocaïne permet d'obtenir un effet analgésique pendant le traitement.
CONTRE-INDICATIONS
· Ne pas injecter REVOLAX SUB-Q Lidocaïne dans le contour des yeux (cercle oculaire ou paupières).
· Ne pas injecter REVOLAX SUB-Q Lidocaïne dans les vaisseaux sanguins (intravasculaire).
· REVOLAX SUB-Q Lidocaïne ne doit pas être utilisé chez :
- Les femmes enceintes ou qui allaitent.
- Les personnes âgées de moins de 18 ans.
- Les patients connus pour leur hypersensibilité à l'acide hyaluronique.
- Les patients qui ont tendance à développer des cicatrices hypertrophiques.
· REVOLAX SUB-Q Lidocaïne ne doit pas être utilisé en association avec une thérapie laser, un peeling chimique ou une abrasion cutanée.
· REVOLAX SUB-Q Lidocaïne ne doit pas être utilisé dans les zones présentant des processus inflammatoires et infectieux cutanés.
· Il convient de prendre en considération la dose totale de lidocaïne administrée si un bloc dentaire ou une administration topique de
lidocaïne est utilisée simultanément. De fortes doses de lidocaïne (plus de 400 mg) peuvent provoquer des réactions toxiques aiguës se manifestant par des symptômes affectant le système nerveux central et la conduction cardiaque.
· La lidocaïne doit être utilisée avec prudence chez les patients recevant d'autres anesthésiques locaux ou des agents structurellement, par
exemple, certains anti-arythmiques, car les effets toxiques systémiques peuvent être additifs.
· La lidocaïne doit être utilisée avec prudence chez les patients souffrant d'épilepsie, d'altération de la conduction cardiaque, d'altération grave de la fonction hépatique ou de dysfonctionnement rénal grave. Les injections péribulbaires d'anesthésiques locaux comportent un faible risque de dysfonctionnement persistant du muscle oculaire.
MISE EN GARDE
REVOLAX SUBQ lidocaïne est uniquement destiné à être utilisé comme implant intradermique.
Assurez-vous que le produit n'est pas périmé et que la stérilité n'a pas été compromise avant l'utilisation. Le produit est à usage unique ; ne
pas le réutiliser.
En cas de réutilisation, cela peut entraîner une diminution des performances du dispositif et une grave infection croisée. Les aiguilles et seringues utilisées doivent être jetées dans un récipient prévu à cet effet.
LES PRÉCAUTIONS D'EMPLOI
REVOLAX SUBQ lidocaïne est conditionné pour un usage unique par patient. Ne pas stériliser à nouveau.
. Ne pas utiliser si l'emballage est ouvert ou endommagé.
· REVOLAX SUBQ Lidocaïne ne doit pas être injecté dans une zone où il y a un implant.
· REVOLAX SUBQ Lidocaïne ne doit pas être mélangé avec d'autres produits avant l'implantation du dispositif.
· Les produits à base d'acide hyaluronique ont une incompatibilité connue avec les sels d'ammonium quaternaire tels que le chlorure de benzalkonium.
· Ne pas l'utiliser chez les patients souffrant de troubles de la coagulation ou chez les patients qui ont suivi un traitement avec des
anticoagulants thrombolytiques ou des inhibiteurs de l'agrégation plaquettaire au cours des 2 semaines précédentes. De même, il est
recommandé d'éviter de prendre de l'aspirine, desanti-inflammatoires non stéroïdiens ou de la vitamine C à forte dose la semaine précédant
l'injection.
· Il est recommandé aux patients de ne pas se maquiller pendant 12 heures après l'injection et d'éviter toute exposition prolongée au soleil, aux UV, ainsi qu'au froid et à la chaleur extrêmes pendant deux semaines après l'injection.
· Si l'aiguille est bloquée, n'augmentez pas la pression sur la tige du piston mais arrêtez l'injection et remplacez l'aiguille.
LES EFFETS INDÉSIRABLES
Les médecins doivent informer les patients qu'il existe des effets indésirables potentiels qui peuvent survenir immédiatement ou être retardés après l'injection. Ces effets indésirables incluent, sans s'y limiter à ceux-ci :
· Des réactions inflammatoires telles que rougeur, gonflement et sensibilité peuvent se produire au point d'injection. Ces réactions peuvent durer deux semaines.
· Des nodules ou des indurations sont également possibles au point d'injection.
· Hématomes.
· Coloration ou décoloration du site d'injection.
· Mauvais ou faible effet de remplissage.
· Des cas de nécrose glabellaire, de formation d'abcès, de granulome et d'hypersensibilité immédiate ou retardée ont été signalés dans la
littérature à la suite de l'administration d'hyaluronique et de l'injection. Il est donc important de prendre en compte ces éventuelles
complications.
Les patients doivent signaler dès que possible à leur médecin les réactions inflammatoires qui persistent pendant plus d'une semaine ou tout
autre effet secondaire qui se développe. Le médecin doit traiter ces réactions de manière appropriée
Tout autre effet secondaire indésirable associé à l'injection de REVOLAX SUBQ lidocaïne doit être signalé au distributeur et/ou au fabricant.


acide hyaluronique injectable à usage médical
COMPOSITION
Lidocaine hydrochloride -------------------------3mg/mL
Acide hyaluronique réticulé ------------- -24mg/mL
(Réticulation = BDDE)
Solution saline tamponnée au phosphate, pH7 -Une seringue contient 1,1mL de REVOLAX SUB-Q Lidocaïne
Injection aiguille ou micro-canule
Usage médical strict
LE DOSAGE ET L'ADMINISTRATION
Avant le traitement, il convient d'évaluer l'aptitude du patient au traitement et ses antécédents médicaux. Les patients doivent être informés des indications de REVOLAX SUBQ lidocaïne ainsi que de ses contre-indications et des effets secondaires potentiels avant leur traitement.
La zone à traiter doit être soigneusement désinfectée et le produit doit être injecté dans des conditions de stérilité. Conservez le produit à température ambiante pendant 30 minutes avant l'injection. Avant l'injection, appuyez sur le piston de l’aiguille jusqu'à ce qu'une petite goutte soit visible à l'extrémité de l'aiguille. Injectez lentement dans le derme en utilisant la technique appropriée, soit la technique de ll'enfilage linéaire, soit une série d'injections ponctuelles avec la seringue fournie. La quantité de REVOLAX SUBQ Lidocaïne à injecter par ride dépendra de sa gravité. Si un blanchiment est observé, c'est-à-dire si la peau devient blanche, l'injection doit être arrêtée immédiatement et la zone doit être massée jusqu'à ce qu'elle retrouve sa couleur normale. Après l'injection, il faut masser légèrement le site de correction pour qu'il épouse le contour des tissus environnants.
La retouche d'une correction entreprise avec REVOLAX SUBQ Lidocaïne doit être effectuée immédiatement ou quelques semaines après la première injection.
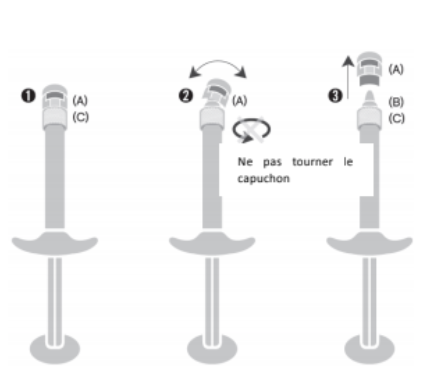
ASSEMBLAGE DE L'AIGUILLE À LA SERINGUE
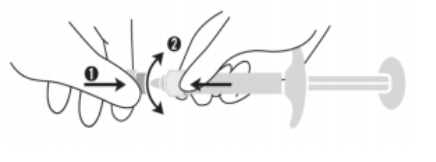
Afin d'utiliser REVOLAX SUBQ Lidocaïne en toute sécurité, il est important que l'aiguille soit correctement assemblée à la seringue. Un
mauvais assemblage peut entraîner la séparation de l'aiguille et de la seringue pendant l'injection.
1. Retirez le capuchon de la seringue
1 Tenez la seringue en haut à droite de la partie (C) du capuchon de l'OVS.
2 De l'autre main, prenez la partie (A) du capuchon et basculez doucement d'avant en
arrière avec précaution jusqu'à ce que le capuchon se déconnecte et puisse être retiré. Ne pas tourner le capuchon.
3 Retirez le capuchon en le tirant droit vers le haut. Ne touchez pas l'embout de la seringue (B)
pour la garder stérilisée.
2. Assemblez une aiguille à la seringue
1 Enfoncez fermement l'aiguille fournie dans la
boîte.
2 Vissez-la doucement dans le sens des aiguilles d'une montre dans la seringue et retirez le capuchon protecteur en tenant le corps de la seringue d'une main, le capuchon protecteur de l'autre, et en tirant les deux mains dans des directions opposées.
Le non-respect de ces précautions peut entraîner un décrochage de l'aiguille et/ou une fuite du produit au niveau du luer-lock.